
IMIQUIMOD- imiquimod cream
Taro Pharmaceutical Indus tries Ltd.
----------
HIGHLIGHTS OF PRESCRIBING INFORMATION
These hig hlights do no t inc lude all the info rmation needed to use Imiquimo d safely and effective ly. See full
prescribing info rmation fo r Imiquimo d.
Imiquimo d Cream, 5%
Fo r to pical use o nly
Initial U.S. Approval: 199 7
INDICATIONS AND USAGE
Imiquimod Cream is indicated for the topical treatment of:
Clinically typical, nonhyperkeratotic, nonhypertrophic actinic ke ratoses (AK) on the face or scalp in immunocompetent
adults (1.1)
External genital and perianal warts/condyloma acuminata in patients 12 years old or older (1.3)
Limitations of Use: Efficacy was not demonstrated for molluscum contagiosum in childre n aged 2-12 (1.4, 8.4)
DOSAGE AND ADMINIST RAT ION
Imiquimod Cream is not for oral, ophthalmic, or intravaginal use .(2)
Actinic ke ratosis: 2 times per week for a full 16 weeks (2.1)
External genital warts (EGW): 3 times per we ek until total clearance or a maximum of 16 weeks (2.3)
DOSAGE FORMS AND STRENGT HS
Imiquimod Cream, 5%, is supplied in single -use packets (24 per box), e ach of which contains 250 mg of the cream,
equivalent to 12.5 mg of imiquimod. (3)
CONTRAINDICATIONS
None (4)
WARNINGS AND PRECAUTIONS
Intense local inflammatory reactions can occur (e.g., skin wee ping, e rosion). Dosing interruption may be require d (2,
5.1, 6)
Severe local inflammatory reactions of the female external ge nitalia can lead to severe vulvar swelling. Se vere vulvar
swelling can lead to urinary retention; dosing should be inte rrupted or discontinued.
Flu-like systemic signs and symptoms including malaise, fe ver, nausea, myalgias and rigors may occur. Dosing
interruption may be required (2, 5.2, 6)
Avoid exposure to sunlight and sunlamps. Wear sunscree n daily (5.3).
Safe ty and e fficacy have not been established for repeat courses of tre atment to the same are a for AK. (5.4)
Treatment of urethral, intra-vaginal, cervical, rectal or intra-anal viral disease is not recommended. (5.6)
Safe ty and e fficacy in immunosuppressed patients have not bee n established (1.5)
ADVERSE REACTIONS
Most common adverse reactions (incidence >28% ) are application site reactions or local skin reactions: itching, burning,
erythema, flaking/scaling/dryness, scabbing/crusting, edema, induration, excoriation, erosion, ulceration. Othe r reporte d
reactions (≥ 1% ) include fatigue, fe ver, and headache (6.1, 6.3)
To repo rt SUSPECT ED ADVERSE REACTIONS, c o ntact the FDA at 1-8 0 0-FDA-108 8 o r www.fda.gov/medwatch.
See 17 for PAT IENT COUNSELING INFORMAT ION and FDA-approved patie nt labeling .
Revised: 7/2011
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Actinic Keratosis
1.3 External Genital Warts
1.4 Limitations of Use
1.5 Unevaluated Populations
2 DOSAGE AND ADMINISTRATION
2.1 Actinic Keratosis
2.3 External Genital Warts
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Local Inflammatory Reactions
5.2 Systemic Reactions
5.3 Ultraviolet Light Exposure
5.4 Unevaluated Uses: Actinic Keratosis
5.6 Unevaluated Uses: External Genital Warts
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience: Actinic Keratosis
6.3 Clinical Trials Experience: External Genital Warts
6.4 Clinical Trials Experience: Dermal Safety Studies
6.5 Postmarketing Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Actinic Keratosis
14.3 External Genital Warts
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
17.1 General Information: All Indications
17.2 Local Skin Reactions: All Indications
17.3 Systemic Reactions: All Indications
17.4 Patients Being Treated for Actinic Keratosis (AK)
17.6 Patients Being Treated for External Genital Warts
*
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Actinic Keratos is
Sections or subsections omitted from the full prescribing information are not listed.

Imiquimod Cream is indicated for the topical treatment of clinically typical, nonhyperkeratotic,
nonhypertrophic actinic keratoses on the face or scalp in immunocompetent adults.
1.3 External Genital Warts
Imiquimod Cream is indicated for the treatment of external genital and perianal warts/condyloma
acuminata in patients 12 years or older.
1.4 Limitations of Use
Imiquimod Cream has been evaluated in children ages 2 to 12 years with molluscum contagiosum and
these studies failed to demonstrate efficacy. [see Use in Specific Populations (8.4)].
1.5 Unevaluated Populations
The safety and efficacy of Imiquimod Cream in immunosuppressed patients have not been established.
Imiquimod Cream should be used with caution in patients with pre-existing autoimmune conditions.
The efficacy and safety of Imiquimod Cream have not been established for patients with Basal Cell
Nevus Syndrome or Xeroderma Pigmentosum.
2 DOSAGE AND ADMINISTRATION
The application frequency for Imiquimod Cream is different for each indication. Imiquimod is not
for oral, ophthalmic, or intravaginal use.
2.1 Actinic Keratos is
Imiquimod Cream should be applied 2 times per week for a full 16 weeks to a defined treatment area on
the face or scalp (but not both concurrently). The treatment area is defined as one contiguous area of
approximately 25 cm (e.g., 5 cm × 5 cm) on the face (e.g. forehead or one cheek) or on the scalp.
Examples of 2 times per week application schedules are Monday and Thursday, or Tuesday and Friday.
Imiquimod Cream should be applied to the entire treatment area and rubbed in until the cream is no
longer visible. No more than one packet of Imiquimod Cream should be applied to the contiguous
treatment area at each application. Imiquimod Cream s hould be applied prior to normal sleeping
hours and left on the s kin for approximately 8 hours, after which time the cream should be
removed by washing the area with mild s oap and water. The prescriber should demonstrate the
proper application technique to maximize the benefit of Imiquimod Cream therapy.
It is recommended that patients wash their hands before and after applying Imiquimod Cream. Before
applying the cream, the patient should wash the treatment area with mild soap and water and allow the
area to dry thoroughly (at least 10 minutes). Contact with the eyes, lips and nostrils should be avoided.
Local skin reactions in the treatment area are common. [see Adverse Reactions (6.1, 6.5)] A rest period of
several days may be taken if required by the patient's discomfort or severity of the local skin reaction.
However, the treatment period s hould not be extended beyond 16 weeks due to miss ed dos es or
res t periods. Response to treatment cannot be adequately assessed until resolution of local skin
reactions. Lesions that do not respond to treatment should be carefully re-evaluated and management
reconsidered.
Imiquimod Cream is packaged in single-use packets, with 24 packets supplied per box. Patients should
be pres cribed no more than 36 packets for the 16-week treatment period. Unused packets should
be discarded. Partially-used packets should be discarded and not reused.
2.3 External Genital Warts
Imiquimod Cream should be applied 3 times per week to external genital/perianal warts. Imiquimod
Cream treatment should continue until there is total clearance of the genital/perianal warts or for a
2

maximum of 16 weeks. Examples of 3 times per week application schedules are: Monday, Wednesday,
Friday or Tuesday, Thursday, Saturday. Imiquimod Cream s hould be applied prior to normal
sleeping hours and left on the s kin for 6-10 hours , after which time the cream should be removed
by was hing the area with mild s oap and water. The prescriber should demonstrate the proper
application technique to maximize the benefit of Imiquimod Cream therapy. It is recommended that
patients wash their hands before and after applying Imiquimod Cream. A thin layer of Imiquimod Cream
should be applied to the wart area and rubbed in until the cream is no longer visible. The application site
should not be occluded. Following the treatment period the cream should be removed by washing the
treated area with mild soap and water.
Local skin reactions at the treatment site are common [see Adverse Reactions (6.3, 6.5)]. A rest period of
several days may be taken if required by the patient's discomfort or severity of the local skin reaction.
Treatment may resume once the reaction subsides. Non-occlusive dressings such as cotton gauze or
cotton underwear may be used in the management of skin reactions.
Imiquimod Cream is packaged in single-use packets which contain sufficient cream to cover a wart area
of up to 20 cm ; use of excessive amounts of cream should be avoided.
3 DOSAGE FORMS AND STRENGTHS
Imiquimod Cream, 5%, is supplied in single-use packets each of which contains 250 mg of the cream,
equivalent to 12.5 mg of imiquimod. Imiquimod Cream is supplied in boxes of 24 packets each.
4 CONTRAINDICATIONS
None.
5 WARNINGS AND PRECAUTIONS
5.1 Local Inflammatory Reactions
Intense local inflammatory reactions including skin weeping or erosion can occur after few applications
of Imiquimod Cream and may require an interruption of dosing. [see Dosage and Administration (2) and
Adverse Reactions (6)]. Imiquimod Cream has the potential to exacerbate inflammatory conditions of the
skin, including chronic graft versus host disease.
Severe local inflammatory reactions of the female external genitalia can lead to severe vulvar swelling.
Severe vulvar swelling can lead to urinary retention. Dosing should be interrupted or discontinued for
severe vulvar swelling.
Administration of Imiquimod Cream is not recommended until the skin is completely healed from any
previous drug or surgical treatment.
5.2 Systemic Reactions
Flu-like signs and symptoms may accompany, or even precede, local inflammatory reactions and may
include malaise, fever, nausea, myalgias and rigors. An interruption of dosing should be considered.
[see Adverse Reactions (6)]
5.3 Ultraviolet Light Expos ure
Exposure to sunlight (including sunlamps) should be avoided or minimized during use of Imiquimod
Cream because of concern for heightened sunburn susceptibility. Patients should be warned to use
protective clothing (e.g., a hat) when using Imiquimod Cream. Patients with sunburn should be advised
not to use Imiquimod Cream until fully recovered. Patients who may have considerable sun exposure,
e.g. due to their occupation, and those patients with inherent sensitivity to sunlight should exercise
caution when using Imiquimod Cream.
2

Imiquimod Cream shortened the time to skin tumor formation in an animal photococarcinogenicity study
[see Nonclinical Toxicology (13.1)]. The enhancement of ultraviolet carcinogenicity is not necessarily
dependent on phototoxic mechanisms. Therefore, patients should minimize or avoid natural or artificial
sunlight exposure.
5.4 Unevaluated Uses : Actinic Keratosis
Safety and efficacy have not been established for Imiquimod Cream in the treatment of actinic keratosis
with repeated use, i.e., more than one treatment course, in the same area.
The safety of Imiquimod Cream applied to areas of skin greater than 25 cm (e.g. 5 cm × 5 cm) for the
treatment of actinic keratosis has not been established [see Clinical Pharmacology (12.3)].
5.6 Unevaluated Uses : External Genital Warts
Imiquimod Cream has not been evaluated for the treatment of urethral, intra-vaginal, cervical, rectal, or
intra-anal human papilloma viral disease.
6 ADVERSE REACTIONS
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed
in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug
and may not reflect the rates observed in practice.
6.1 Clinical Trials Experience: Actinic Keratos is
The data described below reflect exposure to Imiquimod Cream or vehicle in 436 subjects enrolled in
two double-blind, vehicle-controlled studies. Subjects applied Imiquimod Cream or vehicle to a 25 cm
contiguous treatment area on the face or scalp 2 times per week for 16 weeks.
Table 2: Selected Advers e Reactions Occurring in > 1% of Imiquimod-
Treated Subjects and at a Greater Frequency than with Vehicle in the
Combined Studies (Actinic Keratosis )
Preferred Term
Imiquimod Cream
(n=215)
Vehicle (n=221)
Application Site Reaction 71 ( 33%) 32 ( 14%)
Upper Resp Tract Infection 33 ( 15%) 27 ( 12%)
Sinusitis 16 ( 7%) 14 ( 6%)
Headache 11 ( 5%) 7 ( 3%)
Carcinoma Squamous 8 ( 4%) 5 ( 2%)
Diarrhea 6 ( 3%) 2 ( 1%)
Eczema 4 ( 2%) 3 ( 1%)
Back Pain 3 ( 1%) 2 ( 1%)
Fatigue 3 ( 1%) 2 ( 1%)
Fibrillation Atrial 3 ( 1%) 2 ( 1%)
Infection Viral 3 ( 1%) 2 ( 1%)
Dizziness 3 ( 1%) 1 ( <1%)
Vomiting 3 ( 1%) 1 ( <1%)
Urinary Tract Infection 3 ( 1%) 1 ( <1%)
Fever 3 ( 1%) 0 ( 0%)
Rigors 3 ( 1%) 0 ( 0%)
Alopecia 3 ( 1%) 0 ( 0%)
2
2

Table 3: Application Site Reactions Reported by > 1% of Imiquimod-Treated
Subjects and at a Greater Frequency than with Vehicle in the Combined
Studies (Actinic Keratos is )
Included Term
Imiquimod Cream
(n=215)
Vehicle (n=221)
Itching 44 (20%) 17 (8%)
Burning 13 (6%) 4 (2%)
Bleeding 7 (3%) 1 (<1%)
Stinging 6 (3%) 2 (1%)
Pain 6 (3%) 2 (1%)
Induration 5 (2%) 3 (1%)
Tenderness 4 (2%) 3 (1%)
Irritation 4 (2%) 0 (0%)
Local skin reactions were collected independently of the adverse reaction "application site reaction" in
an effort to provide a better picture of the specific types of local reactions that might be seen. The most
frequently reported local skin reactions were erythema, flaking/scaling/dryness, and scabbing/crusting.
The prevalence and severity of local skin reactions that occurred during controlled studies are shown in
the following table.
Table 4: Local Skin Reactions in the Treatment Area as Ass essed by the
Investigator
(Actinic Keratos is)
Imiquimod Cream
(n=215)
Vehicle (n=220)
All
Grades
Severe
All
Grades
Severe
*
Erythema 209 (97%) 38 (18%) 206 (93%) 5 (2%)
Flaking/Scaling/Dryness 199 (93%) 16 (7%) 199 (91%) 7 (3%)
Scabbing/Crusting 169 (79%) 18 (8%) 92 (42%) 4 (2%)
Edema 106 (49%) 0 (0%) 22 (10%) 0 (0%)
Erosion/Ulceration 103 (48%) 5 (2%) 20 (9%) 0 (0%)
Weeping/Exudate 45 (22%) 0 (0%) 3 (1%) 0 (0%)
Vesicles 19 (9%) 0 (0%) 2 (1%) 0 (0%)
The adverse reactions that most frequently resulted in clinical intervention (e.g., rest periods,
withdrawal from study) were local skin and application site reactions. Overall, in the clinical studies,
2% (5/215) of subjects discontinued for local skin/application site reactions. Of the 215 subjects treated,
35 subjects (16%) on Imiquimod Cream and 3 of 220 subjects (1%) on vehicle cream had at least one
rest period. Of these Imiquimod Cream subjects, 32 (91%) resumed therapy after a rest period.
In the AK studies, 22 of 678 (3.2%) of Imiquimod-treated subjects developed treatment site infections
that required a rest period off Imiquimod Cream and were treated with antibiotics (19 with oral and 3
with topical).
Of the 206 Imiquimod subjects with both baseline and 8-week post-treatment scarring assessments, 6
(2.9%) had a greater degree of scarring scores at 8-weeks post-treatment than at baseline.
6.3 Clinical Trials Experience: External Genital Warts
* *
Mild, Moderate, or Severe

In controlled clinical trials for genital warts, the most frequently reported adverse reactions were local
skin and application site reactions.
Some subjects also reported systemic reactions. Overall, 1.2% (4/327) of the subjects discontinued due
to local skin/application site reactions. The incidence and severity of local skin reactions during
controlled clinical trials are shown in the following table.
Table 8: Local Skin Reactions in the Treatment Area as Ass essed by the Inves tigator (External
Genital Warts )
Imiquimod Cream Vehicle
Females Males Females Males
(n=114) (n=156) (n=99) (n=157)
All
Grades
Severe
All
Grades
Severe
All
Grades
Severe
All
Grades
Severe
*
Erythema 74(65%) 4(4%) 90(58%) 6(4%) 21(21%) 0(0%) 34(22%) 0(0%)
Erosion 35(31%) 1(1%) 47(30%) 2(1%) 8(8%) 0(0%) 10(6%) 0(0%)
Excoriation/Flaking 21(18%) 0(0%) 40(26%) 1(1%) 8(8%) 0(0%) 12(8%) 0(0%)
Edema 20(18%) 1(1%) 19(12%) 0(0%) 5(5%) 0(0%) 1(1%) 0(0%)
Scabbing 4(4%) 0(0%) 20(13%) 0(0%) 0(0%) 0(0%) 4(3%) 0(0%)
Induration 6(5%) 0(0%) 11(7%) 0(0%) 2(2%) 0(0%) 3(2%) 0(0%)
Ulceration 9(8%) 3(3%) 7(4%) 0(0%) 1(1%) 0(0%) 1(1%) 0(0%)
Vesicles 3(3%) 0(0%) 3(2%) 0(0%) 0(0%) 0(0%) 0(0%) 0(0%)
Remote site skin reactions were also reported. The severe remote site skin reactions reported for
females were erythema (3%), ulceration (2%), and edema (1%); and for males, erosion (2%), and
erythema, edema, induration, and excoriation/flaking (each 1%). Selected adverse reactions judged to be
probably or possibly related to Imiquimod Cream are listed below.
Table 9: Selected Treatment Related Reactions (External Genital Warts )
Females Males
Imiquimod
Cream
(n=117)
Vehicle
(n=103)
Imiquimod
Cream
(n=156)
Vehicle
(n=158)
*
Application Site Disorders :
Application Site Reactions
Wart Site:
Itching 38(32%) 21(20%) 34(22%) 16(10%)
Burning 30(26%) 12(12%) 14(9%) 8(5%)
Pain 9(8%) 2(2%) 3(2%) 1(1%)
Soreness 3(3%) 0(0%) 0(0%) 1(1%)
Fungal Infection 13(11%) 3(3%) 3(2%) 1(1%)
Sys temic Reactions :
Headache 5(4%) 3(3%) 8(5%) 3(2%)
Influenza-like symptoms 4(3%) 2(2%) 2(1%) 0(0%)
Myalgia 1(1%) 0(0%) 2(1%) 1(1%)
* * * *
Mild, Moderate, or Severe
Incidences reported without regard to causality with Imiquimod Cream.
*

Adverse reactions judged to be possibly or probably related to Imiquimod Cream and reported by more
than 1% of subjects included:
Application Site Disorders : burning, hypopigmentation, irritation, itching, pain, rash, sensitivity,
soreness, stinging, tenderness
Remote Site Reactions: bleeding, burning, itching, pain, tenderness, tinea cruris
Body as a Whole: fatigue, fever, influenza-like symptoms
Central and Peripheral Nervous System Disorders : headache
Gas tro-Intestinal Sys tem Disorders : diarrhea
Mus culo-Skeletal System Disorders : myalgia
6.4 Clinical Trials Experience: Dermal Safety Studies
Provocative repeat insult patch test studies involving induction and challenge phases produced no
evidence that Imiquimod Cream causes photoallergenicity or contact sensitization in healthy skin;
however, cumulative irritancy testing revealed the potential for Imiquimod Cream to cause irritation, and
application site reactions were reported in the clinical studies [see Adverse Reactions (6)].
6.5 Pos tmarketing Experience
The following adverse reactions have been identified during post-approval use of Imiquimod Cream.
Because these reactions are reported voluntarily from a population of uncertain size, it is not always
possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Application Site Disorders : tingling at the application site
Body as a Whole: angioedema
Cardiovas cular: capillary leak syndrome, cardiac failure, cardiomyopathy, pulmonary edema,
arrhythmias (tachycardia, atrial fibrillation, palpitations), chest pain, ischemia, myocardial infarction,
syncope.
Endocrine: thyroiditis
Gas tro-Intestinal Sys tem Disorders : abdominal pain
Hematological: decreases in red cell, white cell and platelet counts (including idiopathic
thrombocytopenic purpura), lymphoma
Hepatic: abnormal liver function
Infections and Infes tations : herpes simplex
Mus culo-Skeletal System Disorders : arthralgia
Neurops ychiatric: agitation, cerebrovascular accident, convulsions (including febrile convulsions),
depression, insomnia, multiple sclerosis aggravation, paresis, suicide
Res piratory: dyspnea
Urinary Sys tem Dis orders : proteinuria, dysuria, urinary retention
Skin and Appendages : exfoliative dermatitis, erythema multiforme, hyperpigmentation, hypertrophic
scar
Vascular: Henoch-Schonlein purpura syndrome
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy

8.1 Pregnancy
Pregnancy Category C:
Note: The Maximum Recommended Human Dose (MRHD) was set at 2 packets per treatment of
Imiquimod Cream (25 mg imiquimod) for the animal multiple of human exposure ratios presented in this
label. If higher doses than 2 packets of Imiquimod Cream are used clinically, then the animal multiple of
human exposure would be reduced for that dose. A non-proportional increase in systemic exposure
with increased dose of Imiquimod Cream was noted in the clinical pharmacokinetic study conducted in
actinic keratosis subjects [see Clinical Pharmacology (12.3)]. The AUC after topical application of 6
packets of Imiquimod Cream was 8 fold greater than the AUC after topical application of 2 packets of
Imiquimod Cream in actinic keratosis subjects.
Therefore, if a dose of 6 packets per treatment of Imiquimod Cream was topically administered to an
individual, then the animal multiple of human exposure would be either 1/3 of the value provided in the
label (based on body surface area comparisons) or 1/8 of the value provided in the label (based on AUC
comparisons). The animal multiples of human exposure calculations were based on weekly dose
comparisons for the carcinogenicity studies described in this label. The animal multiples of human
exposure calculations were based on daily dose comparisons for the reproductive toxicology studies
described in this label.
Systemic embryofetal development studies were conducted in rats and rabbits. Oral doses of 1, 5 and 20
mg/kg/day imiquimod were administered during the period of organogenesis (gestational days 6 – 15) to
pregnant female rats. In the presence of maternal toxicity, fetal effects noted at 20 mg/kg/day (577×
MRHD based on AUC comparisons) included increased resorptions, decreased fetal body weights,
delays in skeletal ossification, bent limb bones, and two fetuses in one litter (2 of 1567 fetuses)
demonstrated exencephaly, protruding tongues and low-set ears. No treatment related effects on
embryofetal toxicity or teratogenicity were noted at 5 mg/kg/day (98× MRHD based on AUC
comparisons).
Intravenous doses of 0.5, 1 and 2 mg/kg/day imiquimod were administered during the period of
organogenesis (gestational days 6 – 18) to pregnant female rabbits. No treatment related effects on
embryofetal toxicity or teratogenicity were noted at 2 mg/kg/day (1.5× MRHD based on BSA
comparisons), the highest dose evaluated in this study, or 1 mg/kg/day (407× MRHD based on AUC
comparisons).
A combined fertility and peri- and post-natal development study was conducted in rats. Oral doses of 1,
1.5, 3 and 6 mg/kg/day imiquimod were administered to male rats from 70 days prior to mating through
the mating period and to female rats from 14 days prior to mating through parturition and lactation. No
effects on growth, fertility, reproduction or post-natal development were noted at doses up to 6
mg/kg/day (87× MRHD based on AUC comparisons), the highest dose evaluated in this study. In the
absence of maternal toxicity, bent limb bones were noted in the F1 fetuses at a dose of 6 mg/kg/day (87×
MRHD based on AUC comparisons). This fetal effect was also noted in the oral rat embryofetal
development study conducted with imiquimod. No treatment related effects on teratogenicity were noted
at 3 mg/kg/day (41× MRHD based on AUC comparisons).
There are no adequate and well-controlled studies in pregnant women. Imiquimod Cream should be used
during pregnancy only if the potential benefit justifies the potential risk to the fetus.
8.3 Nurs ing Mothers
It is not known whether imiquimod is excreted in human milk following use of Imiquimod Cream.
Because many drugs are excreted in human milk, caution should be exercised when Imiquimod Cream is
administered to nursing women.
8.4 Pediatric Us e
AK is not a condition generally seen within the pediatric population. The safety and efficacy of
Imiquimod Cream for AK in patients less than 18 years of age have not been established.

Safety and efficacy in patients with external genital/perianal warts below the age of 12 years have not
been established.
Imiquimod Cream was evaluated in two randomized, vehicle-controlled, double-blind trials involving
702 pediatric subjects with molluscum contagiosum (MC) (470 exposed to Imiquimod; median age 5
years, range 2-12 years). Subjects applied Imiquimod Cream or vehicle 3 times weekly for up to 16
weeks. Complete clearance (no MC lesions) was assessed at Week 18. In Study 1, the complete
clearance rate was 24% (52/217) in the Imiquimod Cream group compared with 26% (28/106) in the
vehicle group. In Study 2, the clearance rates were 24% (60/253) in the Imiquimod Cream group
compared with 28% (35/126) in the vehicle group. These studies failed to demonstrate efficacy.
Similar to the studies conducted in adults, the most frequently reported adverse reaction from 2 studies
in children with molluscum contagiosum was application site reaction. Adverse events which occurred
more frequently in Imiquimod-treated subjects compared with vehicle-treated subjects generally
resembled those seen in studies in indications approved for adults and also included otitis media (5%
Imiquimod vs. 3% vehicle) and conjunctivitis (3% Imiquimod vs. 2% vehicle).
Erythema was the most frequently reported local skin reaction. Severe local skin reactions reported by
Imiquimod-treated subjects in the pediatric studies included erythema (28%), edema (8%),
scabbing/crusting (5%), flaking/scaling (5%), erosion (2%) and weeping/exudate (2%).
Systemic absorption of imiquimod across the affected skin of 22 subjects aged 2 to 12 years with
extensive MC involving at least 10% of the total body surface area was observed after single and
multiple doses at a dosing frequency of 3 applications per week for 4 weeks. The investigator
determined the dose applied, either 1, 2 or 3 packets per dose, based on the size of the treatment area
and the subject's weight. The overall median peak serum drug concentrations at the end of week 4 was
between 0.26 and 1.06 ng/mL except in a 2-year old female who was administered 2 packets of study
drug per dose, had a C of 9.66 ng/mL after multiple dosing. Children aged 2-5 years received doses
of 12.5 mg (one packet) or 25 mg (two packets) of imiquimod and had median multiple-dose peak serum
drug levels of approximately 0.2 or 0.5 ng/mL, respectively.
Children aged 6-12 years received doses of 12.5 mg, 25 mg, or 37.5 mg (three packets) and had median
multiple dose serum drug levels of approximately 0.1, 0.15, or 0.3 ng/mL, respectively.
Among the 20 subjects with evaluable laboratory assessments, the median WBC count decreased by
1.4*10 /L and the median absolute neutrophil count decreased by 1.42*10 /L.
8.5 Geriatric Use
Of the 215 subjects treated with Imiquimod Cream in the AK clinical studies, 127 subjects (59%) were
65 years and older, while 60 subjects (28%) were 75 years and older. No overall differences in safety
or effectiveness were observed between these subjects and younger subjects. No other clinical
experience has identified differences in responses between the elderly and younger subjects, but
greater sensitivity of some older individuals cannot be ruled out.
10 OVERDOSAGE
Topical overdosing of Imiquimod Cream could result in an increased incidence of severe local skin
reactions and may increase the risk for systemic reactions.
The most clinically serious adverse event reported following multiple oral imiquimod doses of >200
mg (equivalent to imiquimod content of >16 packets) was hypotension, which resolved following oral
or intravenous fluid administration.
11 DESCRIPTION
Imiquimod Cream, 5% is an immune response modifier for topical administration.
max
9 9
Each gram contains 50 mg of imiquimod in an off-white oil-in-water vanishing cream base consisting of
benzyl alcohol, cetyl alcohol, glycerin, isostearic acid, methylparaben, polysorbate 60, propylparaben,
purified water, sorbitan monostearate, stearyl alcohol, white petrolatum, and xanthan gum.
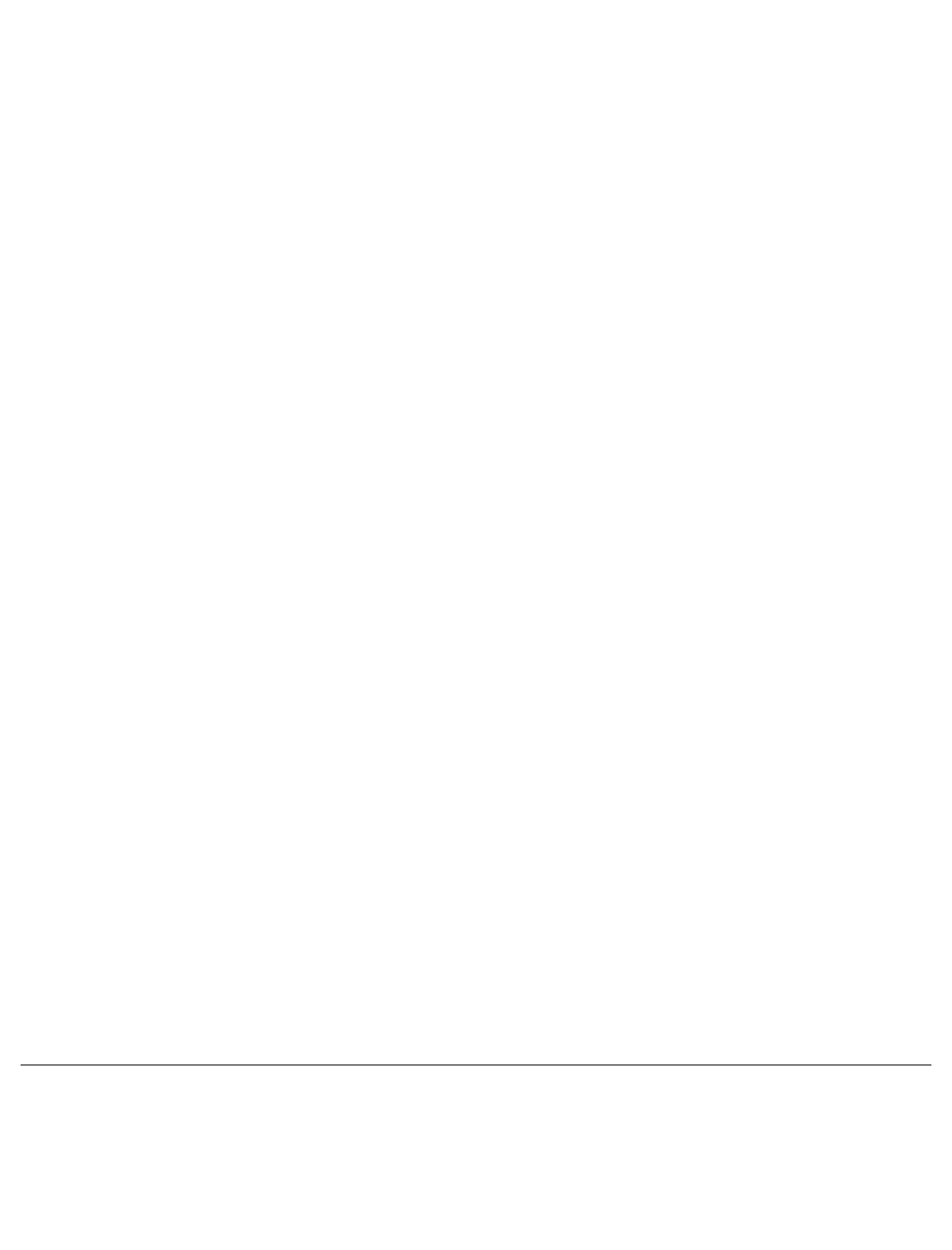
Chemically, imiquimod is 1-(2-methylpropyl)-1H-imidazo[4,5-c]quinolin-4-amine. Imiquimod has a
molecular formula of C H N and a molecular weight of 240.3.
Its structural formula is:
12 CLINICAL PHARMACOLOGY
12.1 Mechanis m of Action
The mechanism of action of Imiquimod Cream in treating AK lesions is unknown.
12.2 Pharmacodynamics
Actinic Keratosis
In a study of 18 subjects with AK comparing Imiquimod Cream to vehicle, increases from baseline in
week 2 biomarker levels were reported for CD3, CD4, CD8, CD11c, and CD68 for Imiquimod Cream
treated subjects; however, the clinical relevance of these findings is unknown.
External Genital Warts
Imiquimod has no direct antiviral activity in cell culture. A study in 22 subjects with genital/perianal
warts comparing Imiquimod Cream and vehicle shows that Imiquimod Cream induces mRNA encoding
cytokines including interferon-α at the treatment site. In addition HPVL1 mRNA and HPV DNA are
significantly decreased following treatment. However, the clinical relevance of these findings is
unknown.
12.3 Pharmacokinetics
Systemic absorption of imiquimod across the affected skin of 58 subjects with AK was observed with a
dosing frequency of 3 applications per week for 16 weeks. Mean peak serum drug concentrations at the
end of week 16 were approximately 0.1, 0.2, and 3.5 ng/mL for the applications to face (12.5 mg
imiquimod, 1 single-use packet), scalp (25 mg, 2 packets) and hands/arms (75 mg, 6 packets),
respectively.
Table 10: Mean Serum Imiquimod Concentration in Adults
Following Adminis tration of the Las t Topical Dose During Week 16
14 16 4

(Actinic Keratos is)
Amount of Imiquimod Cream
applied
Mean peak serum imiquimod
concentration [C ]
12.5 mg (1 packet) 0.1 ng/mL
25 mg (2 packets) 0.2 ng/mL
75 mg (6 packets) 3.5 ng/mL
The application surface area was not controlled when more than one packet was used. Dose
proportionality was not observed. However it appears that systemic exposure may be more dependent
on surface area of application than amount of applied dose. The apparent half-life was approximately 10
times greater with topical dosing than the 2 hour apparent half-life seen following subcutaneous dosing,
suggesting prolonged retention of drug in the skin. Mean urinary recoveries of imiquimod and
metabolites combined were 0.08 and 0.15% of the applied dose in the group using 75 mg (6 packets) for
males and females, respectively following 3 applications per week for 16 weeks.
Systemic absorption of imiquimod was observed across the affected skin of 12 subjects with
genital/perianal warts, with an average dose of 4.6 mg. Mean peak drug concentration of approximately
0.4 ng/mL was seen during the study. Mean urinary recoveries of imiquimod and metabolites combined
over the whole course of treatment, expressed as percent of the estimated applied dose, were 0.11 and
2.41% in the males and females, respectively.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenes is , Mutagenes is , Impairment of Fertility
In an oral (gavage) rat carcinogenicity study, imiquimod was administered to Wistar rats on a 2×/week
(up to 6 mg/kg/day) or daily (3 mg/kg/day) dosing schedule for 24 months. No treatment related tumors
were noted in the oral rat carcinogenicity study up to the highest doses tested in this study of 6 mg/kg
administered 2×/week in female rats (87× MRHD based on weekly AUC comparisons), 4 mg/kg
administered 2×/week in male rats (75× MRHD based on weekly AUC comparisons) or 3 mg/kg
administered 7×/week to male and female rats (153× MRHD based on weekly AUC comparisons). In a
dermal mouse carcinogenicity study, imiquimod cream (up to 5 mg/kg/application imiquimod or 0.3%
imiquimod cream) was applied to the backs of mice 3×/week for 24 months. A statistically significant
increase in the incidence of liver adenomas and carcinomas was noted in high dose male mice compared
to control male mice (251× MRHD based on weekly AUC comparisons). An increased number of skin
papillomas was observed in vehicle cream control group animals at the treated site only. The
quantitative composition of the vehicle cream used in the dermal mouse carcinogenicity study is the
same as the vehicle cream used for Imiquimod Cream, minus the active moiety (imiquimod).
In a 52-week dermal photoco-carcinogenicity study, the median time to onset of skin tumor formation
was decreased in hairless mice following chronic topical dosing (3×/week; 40 weeks of treatment
followed by 12 weeks of observation) with concurrent exposure to UV radiation (5 days per week) with
the Imiquimod Cream vehicle alone. No additional effect on tumor development beyond the vehicle
effect was noted with the addition of the active ingredient, imiquimod, to the vehicle cream.
Imiquimod revealed no evidence of mutagenic or clastogenic potential based on the results of five in
vitro genotoxicity tests (Ames assay, mouse lymphoma L5178Y assay, Chinese hamster ovary cell
chromosome aberration assay, human lymphocyte chromosome aberration assay and SHE cell
transformation assay) and three in vivo genotoxicity tests (rat and hamster bone marrow cytogenetics
assay and a mouse dominant lethal test).
Daily oral administration of imiquimod to rats, throughout mating, gestation, parturition and lactation,
demonstrated no effects on growth, fertility or reproduction, at doses up to 87× MRHD based on AUC
comparisons.
max

14 CLINICAL STUDIES
14.1 Actinic Keratosis
In two double-blind, vehicle-controlled clinical studies, 436 subjects with AK were randomized to
treatment with either Imiquimod Cream or vehicle cream 2 times per week for 16 weeks. The studies
enrolled subjects with 4 to 8 clinically typical, visible, discrete, nonhyperkeratotic, nonhypertrophic
AK lesions within a 25 cm contiguous treatment area on either the face or scalp. The 25 cm
contiguous treatment area could be of any dimensions e.g., 5 cm × 5 cm, 3 cm by 8.3 cm, 2 cm by 12.5
cm. Study subjects ranged from 37 to 88 years of age (median 66 years) and 55% had Fitzpatrick skin
type I or II. All Imiquimod-treated subjects were Caucasians.
On a scheduled dosing day, the study cream was applied to the entire treatment area prior to normal
sleeping hours and left on for approximately 8 hours. Twice weekly dosing was continued for a total of
16 weeks. The clinical response of each subject was evaluated 8 weeks after the last scheduled
application of study cream. Efficacy was assessed by the complete clearance rate, defined as the
proportion of subjects at the 8-week post-treatment visit with no (zero) clinically visible AK lesions in
the treatment area. Complete clearance included clearance of all baseline lesions, as well as any new or
sub-clinical AK lesions which appeared during therapy.
Complete and partial clearance rates are shown in the table below. The partial clearance rate was
defined as the percentage of subjects in whom 75% or more baseline AK lesions were cleared.
Table 11: Clearance Rates (AK)
Complete Clearance Rates (100% AK Lesions Cleared)
Study Imiquimod Cream Vehicle
Study AK1 46% (49/107) 3% (3/110)
Study AK2 44% (48/108) 4% (4/111)
Partial and Complete Clearance Rates (75% or More Bas eline AK Les ions
Cleared)
Study Imiquimod Cream Vehicle
Study AK1 60% (64/107) 10% (11/110)
Study AK2 58% (63/108) 14% (15/111)
Sub-clinical AK lesions may become apparent in the treatment area during treatment with Imiquimod
Cream. During the course of treatment, 48% (103/215) of subjects experienced an increase in AK
lesions relative to the number present at baseline within the treatment area. Subjects with an increase in
AK lesions had a similar response to those with no increase in AK lesions.
14.3 External Genital Warts
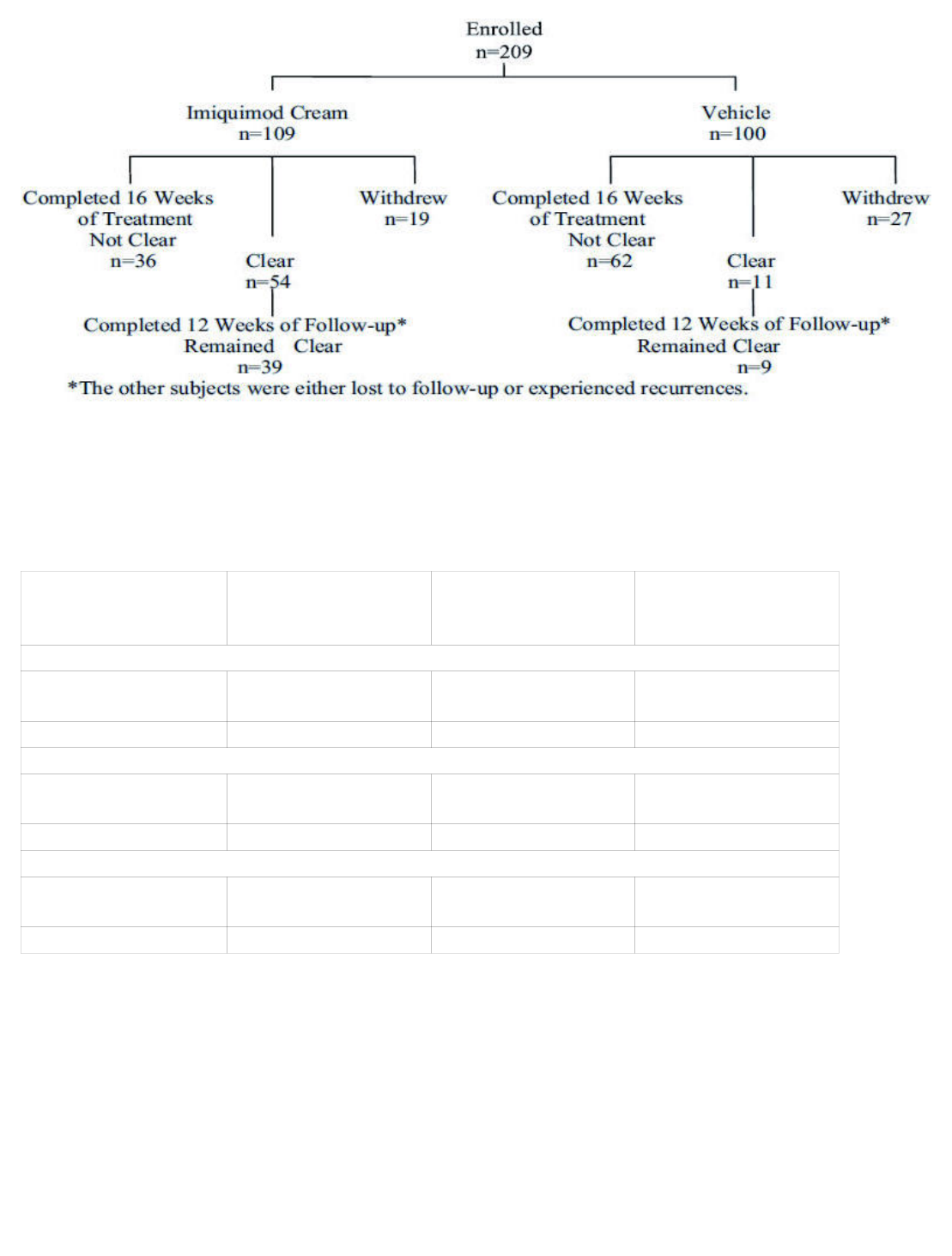
In a double-blind, placebo-controlled clinical study, 209 otherwise healthy subjects 18 years of age and
older with genital/perianal warts were treated with Imiquimod Cream or vehicle control 3 times per
week for a maximum of 16 weeks. The median baseline wart area was 69 mm (range 8 to 5525 mm ).
Subject accountability is shown in the figure below.
Figure 1: Subject Accountability (External Genital Warts )
2 2
2 2
Data on complete clearance are listed in the table below. The median time to complete wart clearance
was 10 weeks.
Table 14: Complete Clearance Rates (External Genital Warts) - Study EGW1
Treatment
Subjects with
Complete Clearance
of Warts
Subjects Without
Follow-up
Subjects with Warts
Remaining at Week
16
Overall
Imiquimod Cream
(n =109)
54 (50%) 19 (17%) 36 (33%)
Vehicle (n =100) 11 (11%) 27 (27%) 62 (62%)
Females
Imiquimod Cream
(n =46)
33 (72%) 5 (11%) 8 (17%)
Vehicle (n =40) 8 (20%) 13 (33%) 19 (48%)
Males
Imiquimod Cream
(n =63)
21 (33%) 14 (22%) 28 (44%)
Vehicle (n =60) 3 (5%) 14 (23%) 43 (72%)
16 HOW SUPPLIED/STORAGE AND HANDLING
Imiquimod Cream, 5%, is supplied in single-use packets which contain 250 mg of the cream:
Box of 24 single-use packets – NDC 51672-4145-6 Discard unused packet.
Store at 4° - 25°C (39° - 77°F)
Avoid freezing.
Keep out of reach of children.
17 PATIENT COUNSELING INFORMATION

See FDA-Approved Patient Labeling
17.1 General Information: All Indications
Imiquimod Cream should be used as directed by a physician [see Dosage and Administration (2)].
Imiquimod Cream is for external use only. Contact with the eyes, lips and nostrils should be avoided.
[see Indications and Usage (1) and Dosage and Administration (2)] The treatment area should not be
bandaged or otherwise occluded. Partially-used packets should be discarded and not reused. The
prescriber should demonstrate the proper application technique to maximize the benefit of Imiquimod
Cream therapy.
It is recommended that patients wash their hands before and after applying Imiquimod cream.
17.2 Local Skin Reactions: All Indications
Patients may experience local skin reactions during treatment with Imiquimod Cream (even with normal
dosing). Potential local skin reactions include erythema, edema, vesicles, erosions/ulcerations,
weeping/exudate, flaking/scaling/dryness, and scabbing/crusting. These reactions can range from mild
to severe in intensity and may extend beyond the application site onto the surrounding skin. Patients may
also experience application site reactions such as itching and/or burning [see Adverse Reactions (6)].
Local skin reactions may be of such an intensity that patients may require rest periods from treatment.
Treatment with Imiquimod Cream can be resumed after the skin reaction has subsided, as determined by
the physician. Patients should contact their physician promptly if they experience any sign or symptom at
the application site that restricts or prohibits their daily activity or makes continued application of the
cream difficult. Because of local skin reactions, during treatment and until healed, the treatment area is
likely to appear noticeably different from normal skin. Localized hypopigmentation and
hyperpigmentation have been reported following use of Imiquimod Cream. These skin color changes
may be permanent in some patients.
17.3 Systemic Reactions: All Indications
Patients may experience flu-like systemic signs and symptoms during treatment with Imiquimod Cream
(even with normal dosing). Systemic signs and symptoms may include malaise, fever, nausea, myalgias
and rigors [see Adverse Reactions (6)]. An interruption of dosing should be considered.
17.4 Patients Being Treated for Actinic Keratos is (AK)
Dosing is 2 times per week for a full 16 weeks, unless otherwise directed by the physician. However,
the treatment period should not be extended beyond 16 weeks due to missed doses or rest periods [see
Dosage and Administration (2.1)].
It is recommended that the treatment area be washed with mild soap and water 8 hours following
Imiquimod Cream application.
Most patients using Imiquimod Cream for the treatment of AK experience erythema,
flaking/scaling/dryness and scabbing/crusting at the application site with normal dosing [see Adverse
Reactions (6.1)].
Use of sunscreen is encouraged, and patients should minimize or avoid exposure to natural or artificial
sunlight (tanning beds or UVA/B treatment) while using Imiquimod Cream [see Warnings and Precautions
(5.3)].
Sub-clinical AK lesions may become apparent in the treatment area during treatment and may
subsequently resolve [see Clinical Studies (14.1)].
17.6 Patients Being Treated for External Genital Warts
Dosing is 3 times per week to external genital/perianal warts. Imiquimod Cream treatment should
continue until there is total clearance of the genital/perianal warts or for a maximum of 16 weeks.
It is recommended that the treatment area be washed with mild soap and water 6-10 hours following
Imiquimod Cream application.
It is common for patients to experience local skin reactions such as erythema, erosion,
excoriation/flaking, and edema at the site of application or surrounding areas. Most skin reactions are
mild to moderate.
Sexual (genital, anal, oral) contact should be avoided while Imiquimod Cream is on the skin.
Application of Imiquimod Cream in the vagina is considered internal and should be avoided. Female
patients should take special care if applying the cream at the opening of the vagina because local skin
reactions on the delicate moist surfaces can result in pain or severe swelling, and may cause difficulty
in passing urine or inability to urinate. Uncircumcised males treating warts under the foreskin should
retract the foreskin and clean the area daily.
New warts may develop during therapy, as Imiquimod Cream is not a cure.
The effect of Imiquimod Cream on the transmission of genital/perianal warts is unknown. Imiquimod
Cream may weaken condoms and vaginal diaphragms; therefore, concurrent use is not recommended.
Should severe local skin reaction occur, the cream should be removed by washing the treatment area
with mild soap and water.
Rx Only
Manufactured by:
Taro Pharmaceutical Industries, Ltd., Haifa Bay, Israel 26110
Distributed by:
Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532
20023-0111-0
Patient Information
Imiquimod
Cream, 5%
Important: For use on the s kin only (topical). Do not us e Imiquimod Cream in or near your
mouth, eyes , nos e or vagina.
Read the Patient Information that comes with Imiquimod Cream before you start using it and each time
you get a refill. There may be new information. This leaflet does not take the place of talking with your
healthcare provider about your medical condition or treatment. If you do not understand the information,
or have any questions about Imiquimod Cream, talk with your healthcare provider or pharmacist.
What is Imiquimod Cream?
Imiquimod Cream is a prescription medicine for use on the skin only (topical) to treat:
actinic keratosis on the face or scalp in adults with a normal immune system. Actinic keratosis is
caused by too much sun exposure.
warts on or around the genitals or anus in people 12 years and older.
Imiquimod Cream will not cure your genital or perianal warts. New warts may develop during treatment
with Imiquimod Cream. It is not known if Imiquimod Cream can stop you from spreading genital or
perianal warts to other people. It is not known if Imiquimod Cream is safe and effective in:
people who do not have a normal immune system.
the treatment of xeroderma pigmentosum.
the treatment of actinic keratosis with more than one treatment course in the same affect area.
It is not known if Imiquimod Cream is safe and effective in children younger than 18 years of age for the
treatment of actinic keratosis. Children usually do not get actinic keratoses.
It is not known if Imiquimod Cream is safe and effective in children younger than 12 years of age for the
treatment of genital and perianal warts.
What s hould I tell my healthcare provider before using Imiquimod Cream?
Before us ing Imiquimod Cream, tell your healthcare provider if you:
have problems with your immune system.
are being treated or have been treated for actinic keratosis with other medicines or surgery. You
should not use Imiquimod Cream until you have healed from other treatments.
have any other skin problems.
have any other medical conditions.
are pregnant or planning to become pregnant. It is not known if Imiquimod Cream can harm your
unborn baby. Talk to your healthcare provider if you are pregnant or plan to become pregnant.
are breastfeeding or plan to breastfeed. It is not known if Imiquimod Cream passes into your breast
milk and if it can harm your baby. Talk to your healthcare provider about the best way to feed your
baby if you use Imiquimod Cream.
Tell your healthcare provider about all the medicines you take, including prescription and non-
prescription medicines, vitamins and herbal supplements.
Especially tell your healthcare provider if you have had other treatments for genital warts or warts
around your anus, or actinic keratosis. Imiquimod Cream should not be used until your skin has healed
from other treatments.
Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist
when you get a new medicine.
How should I use Imiquimod Cream?
Do not get Imiquimod Cream in or near your mouth, eyes, nose or vagina.
Use Imiquimod Cream exactly as your healthcare provider tells you to use it. Your healthcare
provider will tell you where to apply Imiquimod Cream and how often and for how long to apply it
for your condition.
Imiquimod Cream is used for different skin conditions. Use Imiquimod Cream only on the area of
your body to be treated. Do not apply Imiquimod Cream to other areas.
Do not use Imiquimod Cream longer than prescribed. Using too much Imiquimod Cream, or using it
too often, or for too long can increase your chances for having a severe skin reaction or other side
effect.
You should follow-up with your healthcare provider regularly for check-ups while using
Imiquimod Cream.
Talk to your healthcare provider if you think Imiquimod Cream is not working for you.
Applying Imiquimod Cream
Imiquimod Cream should be applied just before your bedtime.
Wash the area where the cream will be applied with mild soap and water. Uncircumcised males
treating warts under their penis foreskin must pull their foreskin back and clean the area before
treatment, and clean the area daily during treatment.
Allow the area to dry for at least 10 minutes.
Wash your hands.
Open a new packet of Imiquimod Cream.
Apply a thin layer of Imiquimod Cream only to the affected area. Do not use more Imiquimod Cream
than is needed to cover the affected area.

Rub the cream into your skin until you can not see the Imiquimod Cream. After applying Imiquimod
Cream, wash your hands well.
Leave the cream on the treated area for the amount of time your healthcare provider tells you. The
length of time that Imiquimod Cream is left on the skin is different for each skin condition that
Imiquimod Cream is used to treat. Do not take a bath or get the treated area wet during this time.
After the right amount of time has passed, wash the treated area with mild soap and water.
If you get Imiquimod Cream in your mouth or in your eyes, rinse well with water right way.
What s hould I avoid while us ing Imiquimod Cream?
Do not cover the treated area with bandages or other closed dressings.
Do not use sunlamps or tanning beds, and avoid sunlight as much as possible during treatment with
Imiquimod Cream. Use sunscreen and wear protective clothing if you go outside during daylight.
Do not have sexual contact including genital, anal, or oral sex when Imiquimod Cream is on your
genital or the skin around your anus. Imiquimod Cream may weaken condoms and vaginal
diaphragms. This means they may not work as well to prevent pregnancy.
What are the pos s ible side effects of Imiquimod Cream?
Imiquimod Cream may cause s erious s ide effects including:
Local skin reactions, including:
skin drainage (weeping)
ulcers
severe swelling near the vagina. This may lead to pain or trouble passing urine or cause you not
to be able to urinate. Female patients should take special care when applying Imiquimod Cream at
the opening of the vagina.
Flu-like s ymptoms : tiredness, fever, nausea, muscle pain and chills.
Tell your healthcare provider right away if you have any of the s ymptoms lis ted above.
The most common side effects of Imiquimod cream include:
itching
burning
redness
flaking and scaling
dryness
scabbing and crusting
swelling
skin that becomes hard or thickened
sores, blisters, or ulcers
changes in skin color that do not always
go away
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of Imiquimod Cream. For more information, ask your
healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-
FDA-1088
How do I s tore Imiquimod Cream?
Store Imiquimod Cream between 39°F to 77°F (4°C to 25°C).
Do not freeze.
Safely throw away unused Imiquimod Cream or partially used Imiquimod cream packets that you do
not need.
Keep Imiquimod Cream and all medicines out of the reach of children.

General information about the s afe and effective us e of Imiquimod Cream
Medicines are sometimes prescribed for purposes other than those listed in this Patient Information
leaflet. Do not use Imiquimod Cream for a condition for which it was not prescribed. Do not give
Imiquimod Cream to other people, even if they have the same symptoms you have. It may harm them.
This Patient Information leaflet summarizes the most important information about Imiquimod Cream. If
you would like more information, talk with your healthcare provider. You can ask your pharmacist or
healthcare provider for information about Imiquimod Cream that is written for the healthcare
professionals.
What are the ingredients in Imiquimod Cream?
Active ingredient: imiquimod Inactive ingredients: benzyl alcohol, cetyl alcohol, glycerin, isostearic
acid, methylparaben, polysorbate 60, propylparaben, purified water, sorbitan monostearate, stearyl
alcohol, white petrolatum, and xanthan gum.
Manufactured by:
Taro Pharmaceutical Industries, Ltd., Haifa Bay, Israel 26110
Distributed by:
Taro Pharmaceuticals U.S.A., Inc., Hawthorne, NY 10532
Revised: January 2011
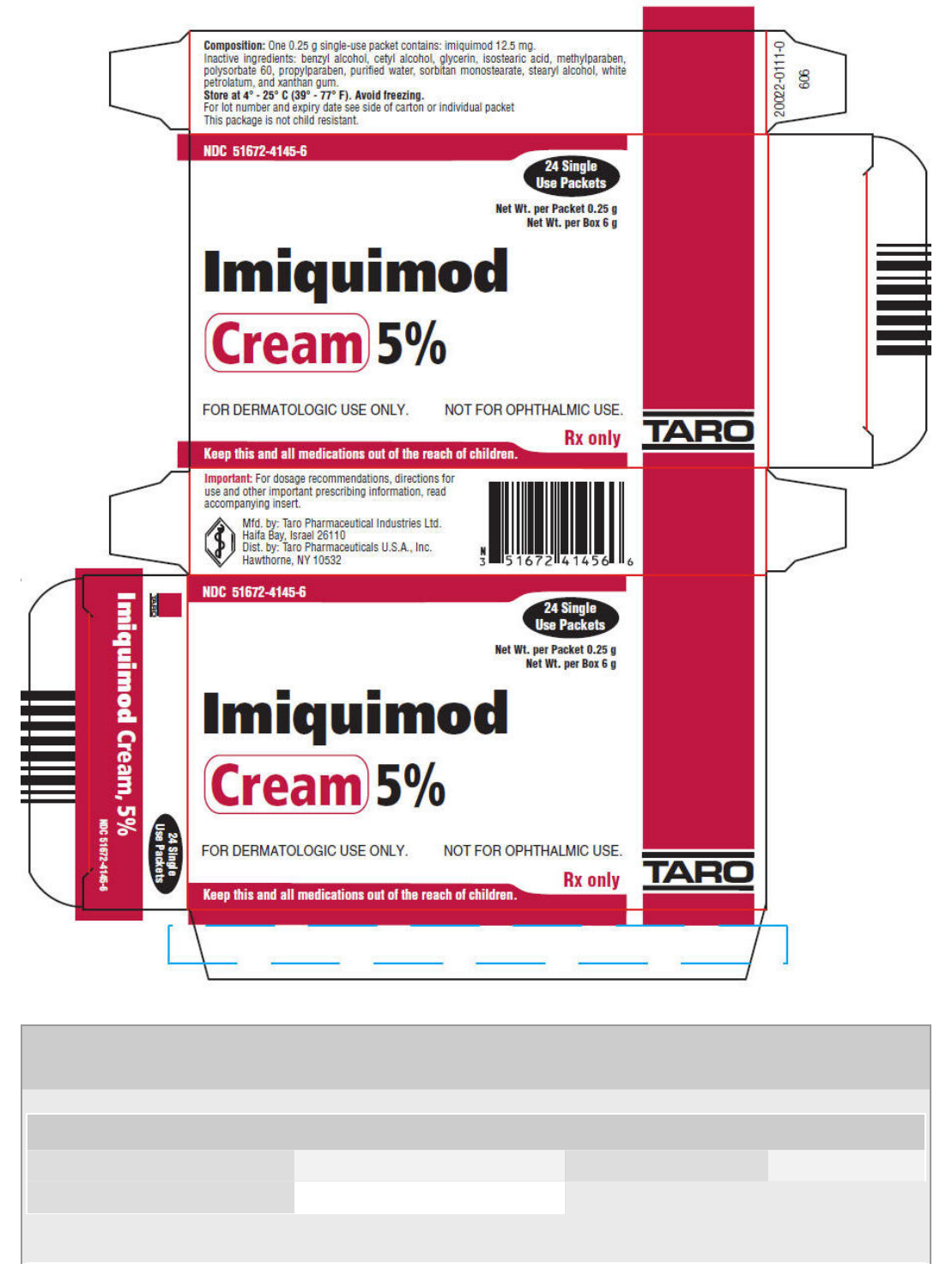
PRINCIPAL DISPLAY PANEL - 24 Packet Carton
NDC 51672-4145-6
24 Single
Use Packets
Net Wt. per Packet 0.25 g
Net Wt. per Box 6 g
Imiquimod
Cream 5%
FOR DERMATOLOGIC USE ONLY.
NOT FOR OPHTHALMIC USE.
Rx only
Keep this and all medications out of the reach of children.
TARO
IMIQUIMOD
imiquimod cream
Product Information
Prod uct T yp e
HUMAN PRESCRIPTION DRUG
Ite m Cod e (S ource )
NDC:52549 -4145
Route of Administration
TOPICAL

Taro Pharmaceutical Industries Ltd.
Active Ingredient/Active Moiety
Ing redient Nam e Basis of Stre ng th Streng th
Imiquimo d (UNII: P1QW714R7M) (Imiquimo d - UNII:P1Q W714R7M) Imiquimo d 50 mg in 1 g
Inactive Ingredients
Ing redient Nam e Streng th
be nz yl a lco ho l (UNII: LKG8 49 4WBH)
ce tyl a lc o ho l (UNII: 9 36 JST6 JCN)
g lyce rin (UNII: PDC6 A3C0 O X)
iso stea ric a cid (UNII: X33R8 U0 0 6 2)
me thylpa ra be n (UNII: A2I8C7HI9 T)
po lyso rba te 6 0 (UNII: CAL22UVI4M)
pro pylpa ra ben (UNII: Z8 IX2SC1OH)
wa ter (UNII: 059 Q F0 KO 0 R)
so rbitan mo no stea ra te (UNII: NVZ4I0 H58 X)
stea ryl a lc o ho l (UNII: 2KR8 9 I4H1Y)
pe tro la tum (UNII: 4T6 H12BN9 U)
xa ntha n gum (UNII: TTV12P4NEE)
Product Characteristics
Color
WHITE
Score
Shap e Siz e
Flavor Imp rint Cod e
Contains
Packaging
# Item Co de Packag e De scriptio n Marketing Start Date Marketing End Date
1 NDC:52549 -4145-6 24 in 1 CARTO N
1 NDC:52549 -4145-8 0 .25 g in 1 PACKET
Marketing Information
Marke ting Cate gory Ap p lication Numb e r or Monograp h Citation Marke ting S tart Date Marke ting End Date
ANDA ANDA20 0 173 0 4/15/20 11
Labeler - T aro Pharmaceutical Indus tries Ltd. (600072078)
Establishment
Name Add re ss ID/FEI Busine ss Op e rations
Taro Pharma ce utica l Industries Ltd. 6 0 0 0 720 78 MANUFACTURE
Revised: 7/2011
